Our Pipeline
Our lead program selectively inhibits the activity of a protein complex called target of rapamycin complex 1, or TORC1, an evolutionarily conserved pathway that contributes to the decline in function of multiple aging organ systems. Our lead product candidate is a TORC1 inhibitor called RTB101.
We are developing RTB101 alone and in combination with other TORC1 inhibitors for multiple aging-related diseases. In addition to developing our TORC1 product candidates, we are building a pipeline of products targeting multiple mechanisms underlying the biology of aging.
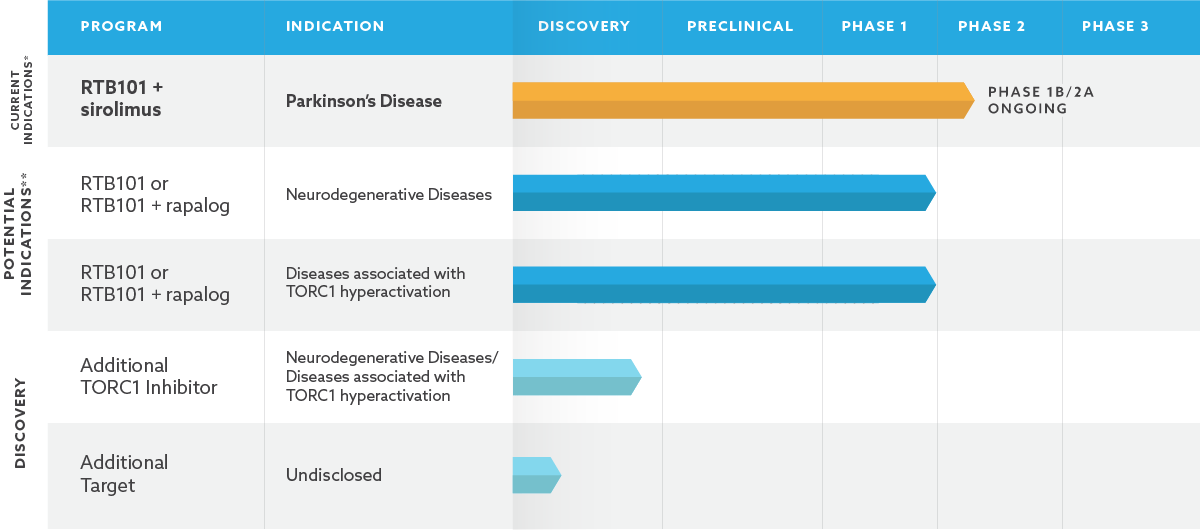
** For neurodegenerative diseases and diseases associated with TORC1 hyperactivation, subject to review by the U.S. Food and Drug Administration, we believe we may have the ability to initiate Phase 2 clinical trials without the need to conduct additional Phase 1 trials.
About RTB101
RTB101 inhibits the phosphorylation of multiple targets downstream of TORC1. Inhibition of TORC1 has been observed to extend lifespan and healthspan in aging preclinical species and to improve the function of multiple organ systems, including neurologic function, suggesting potential benefits in several aging-related diseases.
We currently have a Phase 1b/2a program for RTB101 alone and in combination with sirolimus in Parkinson’s disease. We expect to develop RTB101 for additional indications in other aging-related diseases.
About Parkinson’s Disease
The incidence of PD increases rapidly in people 60 years of age and older, with a mean age at diagnosis of 70.5 years. In PD, neurodegeneration is thought to be caused, at least in part, by the accumulation of aggregates of a protein called alpha-synuclein that are toxic to neurons in the brain. Patients with PD develop shaking, rigidity, slowness of movement and difficulty walking.
Selective and broad inhibition of TORC1 has been shown to ameliorate neurodegenerative disease in several preclinical studies, including models of PD. TORC1 inhibition with RTB101 in combination with sirolimus, another TORC1 inhibitor, may provide a therapeutic benefit to PD patients by potentially inducing autophagy to clear the toxic alpha-synuclein aggregates in neurons.
We are transiting into a blog that focus on beauty product review.
Do check out our Popular Skincare REVIEWS below
- Estee lauder advanced night repair ingredients
- Hanasui vitamin c serum ingredients
- Bio oil ingredients
- Eucerin aquaphor healing ointment ingredients
- Acnes vitamin c serum ingredients
- Bepantol Baby ingredients
- Natur e anti aging eye cream ingredients
- il makiage woke up like this ingredients
- Sheer joy sunscreen ingredients
- Olaplex no 4 shampoo ingredients
- Avoskin-miraculous-refining-toner ingredients
- TRESemmé hairspray ingredients
- e-l-f-poreless-putty-primer ingredients
- Ivory original scent bar soap ingredients
- Cetaphil gentle-skin cleanser ingredients
- Olay regenerist micro-sculpting cream ingredients
- Janssen retin a tretinoina crema ingredients
- Bayer bepantol derma ingredients
- Aveeno daily moisturizing lotion ingredients
- Vaseline protecting jelly ingredients
- Okeeffes working hands hand-cream ingredients
- Kiehls ultra facial cream ingredients
- Aloe pura hudgel aloe-vera ingredients
- Ponds dry-skin cream ingredients
- Eucerin original healing cream ingredients
- Cerave moisturizing cream ingredients
- Cetaphil moisturizing lotion ingredients
- Avon skin so soft original bath oil spray ingredients
- Mederma advanced scar gel ingredients
- Ponds dry-skin cream ingredients
